
The law of multiple proportions states that if two elements form more than one compound between them, the masses of one element combined with a fixed mass of the second element form in ratios of small integers.\)) More important, by measuring the extent of the deflection of the cathode rays in magnetic or electric fields of various strengths, Thomson was able to calculate the mass-to-charge ratio of the particles. This atomic theory is mainly based on the laws of chemical combination. This theory is also known as Dalton’s atomic theory of matter. Atoms are indivisible and indestructible. Discovery of isotopes revealed that the atoms of the same element possess different weights. In 1808, An English scientist John Dalton proposed the first scientific theory of matter. Daltons Atomic Theory 1) All matter is made of atoms. Thus the assumption of Dalton’s theory that the atom is not divisible is false. Dalton’s atomic theory was proposed in 1804 and was the first attempt to describe matter in terms of atoms. 
Daltons Atomic Theory The main points of Daltons atomic theory are: Everything is composed of atoms, which are the indivisible building blocks of matter and cannot be destroyed. Of course, we just as easily could have illustrated the law by considering the mass of nitrogen that combines with one gram of oxygen it works both ways! Limitations of Dalton’s Atomic Theory: Now it is proved that the atom consists of particles like electrons, protons, and neutrons. This image from Daltons A New System of Chemical Philosophy, published in 1808, depicts various atoms and molecules.Note that just as the law of multiple proportions says, the weight of oxygen that combines with unit weight of nitrogen work out to small integers there is a typo in Line 3-where there is a 3, there should be a 4. Atoms of different elements differ in size, mass, and other properties. Atoms of a given element are identical in size, mass, and other properties. Atoms cannot be subdivided, created, or destroyed. The general tenets of this theory are: All matter is composed of extremely small particles called atoms. Line 3 is obtained by dividing the figures of the Line 2 by the smallest O:N ratio in Line 2 (which is the ratio for N 2O). Dalton proposed his atomic theory in 1804. 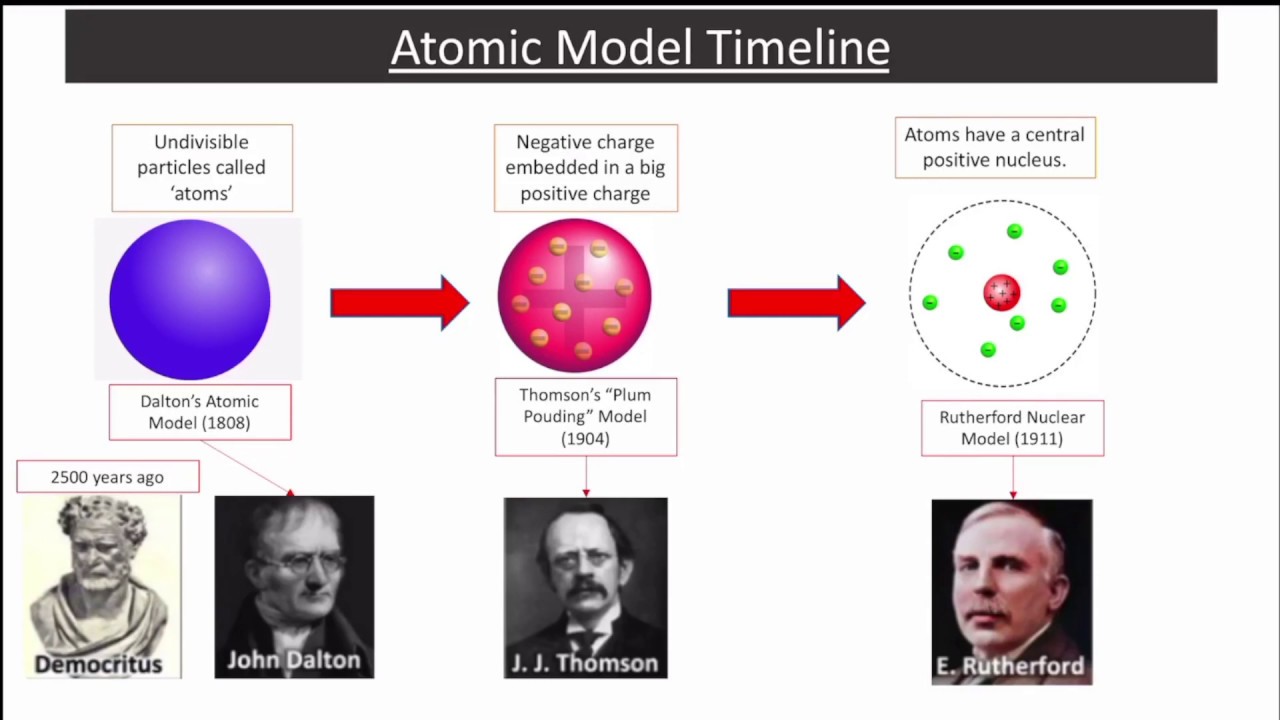
(3) Atoms can neither be created nor destroyed. (2) All atoms of the same element are identical different elements have different types of atom. It involves the following postulates: (1) Elements consist of indivisible small particles (atoms). E) that an atom is predominantly empty space. A theory of chemical combination, first stated by John Dalton in 1803. D) that energy is neither created nor destroyed during a chemical reaction. C) that the properties of matter are determined by the properties of atoms. B) that matter is composed of small indestructible particles. (3) Atoms of different elements have different masses, sizes and also possess different chemical properties. (2) Atoms of the same element are identical in all respect i.e. 
(1) Matter is made up of extremely small indivisible particles called atoms. He was a British physicist, chemist, and meteorologist who is well known for many of his contributions to the pioneering research of atoms, the law of partial pressures, Daltonism, etc. Daltons Atomic Theory states A) that all elements have several isotopes. John Dalton in 1808 put forward theory known as Dalton’s atomic theory. But someone who depends solely on experiment would work these out by finding the mass of O that combines with unit mass (1 g) of nitrogen. The introduction to the early theory of the atom was done by a scientist named John Dalton (1766-1844). The numbers in Line 2 are just the mass ratios of O:N, found by dividing the corresponding ratios in line 1.(These numbers were not known in the early days of Chemistry because atomic weights of most elements were not reliably known.) The law of multiple proportions states that when two elements react to form more than one compound, a fixed mass of one element will react with masses of the other element in a ratio of small, whole numbers. Matter is composed of exceedingly small particles called atoms. These ratios were calculated by simply taking the molar mass of each element, and multiplying by the number of atoms of that element per mole of the compound. Here are the postulates of Dalton’s atomic theory. Line 1 shows the ratio of the relative weights of the two elements in each compound.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
